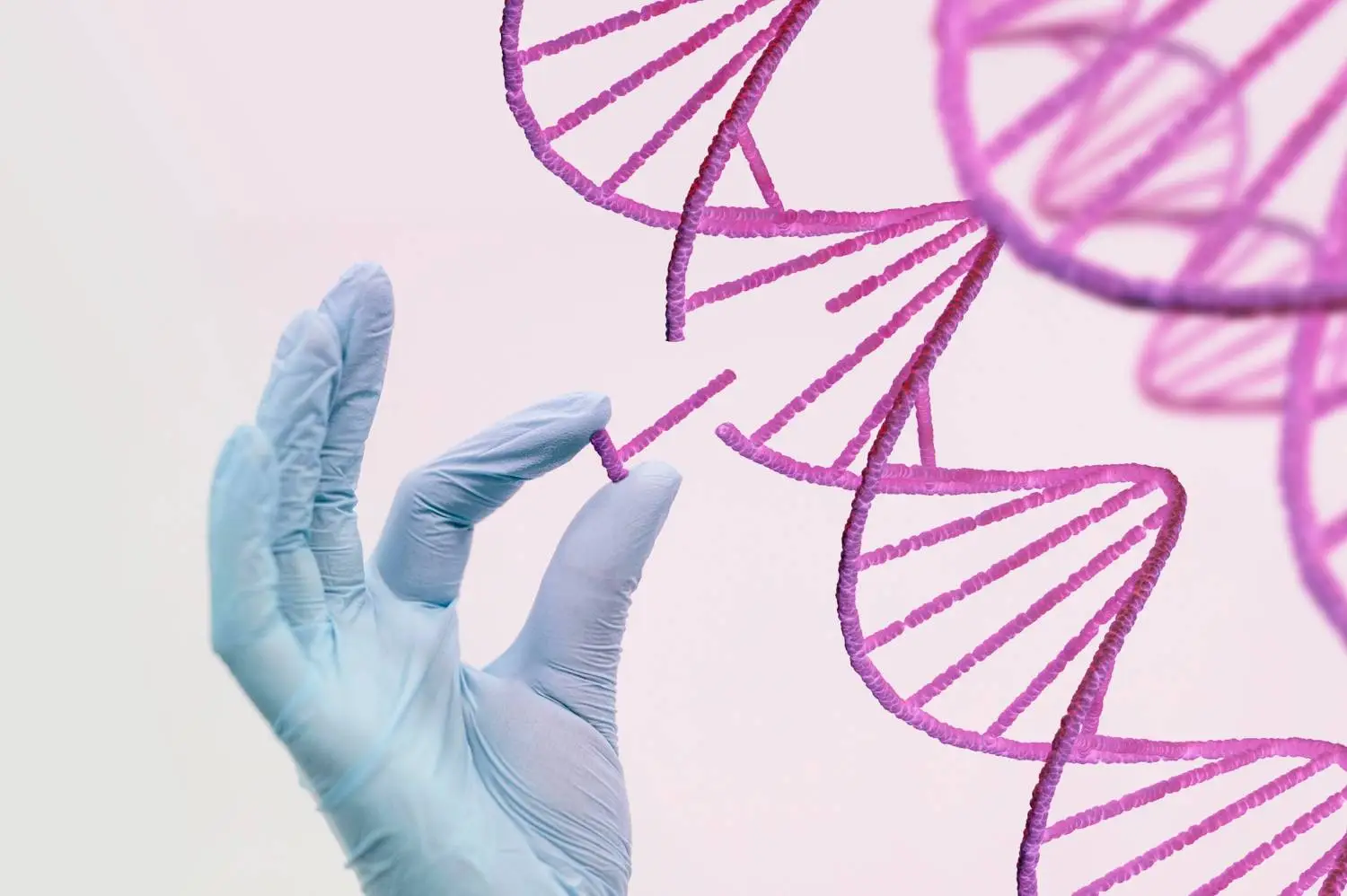
The UK has achieved a historic milestone by becoming the first country to authorize a revolutionary new treatment for two serious inherited blood conditions – sickle cell disease and beta-thalassemia. The cutting-edge therapy utilizes CRISPR gene editing technology to precisely correct errors in patients’ blood stem cells, potentially curing them of debilitating symptoms.
Announced earlier this week, the treatment called Casgevy (exagamglogene autotemcel) has been approved for use by the Medicines and Healthcare Products Regulatory Agency (MHRA). This landmark decision opens the door for this powerful gene editing approach to start benefiting eligible patients after final assessments by the National Institute for Health and Care Excellence (NICE).
Addressing a Major Clinical Need
Casgevy targets two blood disorders sharing an underlying mutation that reduces normal hemoglobin levels. Sickle cell disease distorts red blood cells into inflexible sickled shapes, causing severe pain, organ damage, and shortened lifespan. An estimated 15,000 people in the UK live with this painful condition.
Beta thalassemia patients also face severe anemia, requiring frequent blood transfusions and iron reduction therapy. Bone marrow transplants can cure both diseases but involve significant risks and limited donor availability. Casgevy provides a safer alternative with impressive clinical durability by precisely correcting the original mutations.
This is a world-first and a significant moment for researchers, clinicians and, most of all, people with sickle cell disease and beta thalassaemia.
Professor Josu de la Fuente
Imperial College Healthcare NHS Trust and Imperial College London
How CRISPR Gene Editing Powers the Treatment
Casgevy leverages the Nobel prize-winning CRISPR-Cas9 system, which functions as programmable “molecular scissors” to edit DNA sequences. After harvesting blood stem cells from patients via a standard procedure, scientists use CRISPR in the lab to deactivate the faulty hemoglobin-producing gene.
The edited cells are then infused back into patients, allowing the body to start naturally producing normal hemoglobin. In this autologous cell therapy approach, patients receive their own cells back, avoiding the need for immune suppression.
Remarkable Trial Outcomes for Patients
In recent global clinical trials of Casgevy sponsored by pharma giants Vertex and CRISPR Therapeutics, results have been highly promising:
- 28 out of 29 sickle cell patients became nearly pain-free
- 39 out of 42 beta-thalassemia patients stopped needing transfusions – benefits lasting over a year so far
Such remarkable outcomes have created tremendous excitement about finally having definitive treatments for these chronic disorders via genetic correction. If assessments by NICE also prove favorable, the NHS aims to offer CRISPR therapy like Casgevy to all eligible patients waiting for innovative therapies.
A Glimpse into the Gene Editing Future
By harnessing the power of CRISPR editing applied to stem cells, Casgevy provides a template for treating numerous genetic diseases safely and durably. Its approval heralds wider adoption of next-generation gene and cell therapies, transforming how we combat conditions once deemed incurable.
As personalized medicine continues advancing, we inch closer to a world where genetic screening can catch disorders early when they are most amenable to correction by molecular tools like CRISPR. Bolstered by crucial support infrastructure and policies emphasizing equitable access, the prospects for improving countless lives through technologies like Casgevy continue brightening.
True to its history of pioneering biomedicine advancement, the UK has etched its name in history books as the first regulator globally to embrace CRISPR’s monumental therapeutic potential. But many nations will undoubtedly follow its lead, propelling a new era of genomic and cell medicines.
Story Source: Reference Article
Learn More:
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar










Well done