Researchers from McGill University collaborated with the Canadian Light Source (CLS) at the University of Saskatchewan to unlock the full potential of small biological machinery that can significantly influence human health.
Nonribosomal peptide synthetases (NRPSs), referred to as machines, are large modular enzymes that synthesize secondary metabolites and natural products essential for the production of therapeutic compounds present in a wide range of medications, from antibiotics to immunosuppressants.
According to Camille Fortinez, a recent Ph.D. graduate of McGill’s Department of Biochemistry and lead author of the research published in Nature Communications, these NRPS machines are found in bacteria and fungi, and they are responsible for producing compounds that are used as medications and therapeutics.
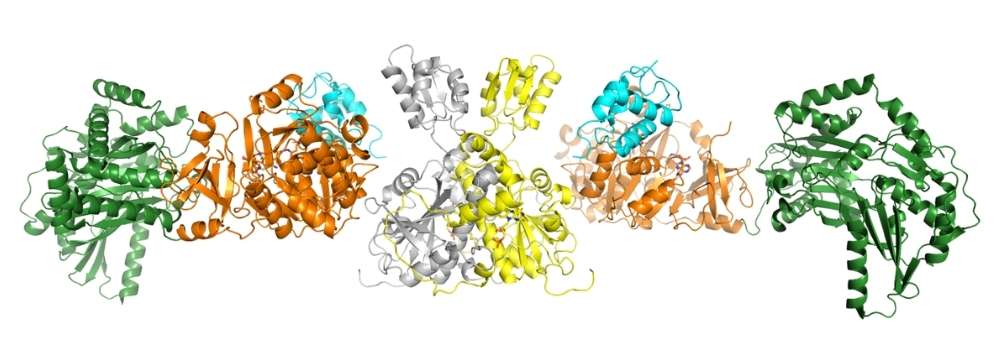
Image Source: https://www.lightsource.ca/public/news/2021-22-q4-jan-march/tiny-machines-in-bacteria-could-help-make-new-medicines.phpCrystal structure of the NRPS complex.
Researchers will be able to design new drugs and combat global health issues like antibiotic resistance by better understanding how NRPSs create therapeutics.
Martin Schmeing, senior author of the paper and Professor of Biochemistry at McGill, and Fortinez examined one common NRPS machine in great detail using the CLS’s CMCF beamline.
“CLS has allowed us to get a really high-resolution diffraction of our NRPS crystals,” says Fortinez. “This high resolution is really integral for allowing us to answer questions and better understand the NRPS.”
The researchers looked at an NRPS common in many bacteria that aids in the production of a toxin that kills algae. Fortinez and Schmeing revealed that a different enzyme is in charge of a critical stage in creating this algae-killing chemical during the process.
The team’s NRPS machines could aid in developing novel treatments, and the algae-killing substance could be tweaked to destroy microorganisms that are harmful to our health. The researchers are optimistic that the detailed data they gathered would aid their efforts.
“The CLS is a wonderful national resource that we are deeply indebted to and it’s a really important resource to keep Canadian science doing as well as we are doing,” told Schmeing.
Story Source: Fortinez, C.M., Bloudoff, K., Harrigan, C. et al. Structures and function of a tailoring oxidase in complex with a nonribosomal peptide synthetase module. Nat Commun 13, 548 (2022). https://doi.org/10.1038/s41467-022-28221-y https://www.lightsource.ca/public/news/2021-22-q4-jan-march/tiny-machines-in-bacteria-could-help-make-new-medicines.php
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar











