Scientists have developed ERX-41, a potent therapeutic drug with a well-defined molecular target (Lysosomal Acid Lipase, LAL) and mechanism of action (disruption of protein folding and production of ER stress) that could be useful in the treatment of patients with multiple solid cancers (Hard to Treat Cancers).
Scientists from the University of Texas, Dallas synthesized a novel chemical that kills a wide range of hard to treat cancers, including triple-negative breast cancer, by exploiting a weakness in cells not previously targeted by other medications.
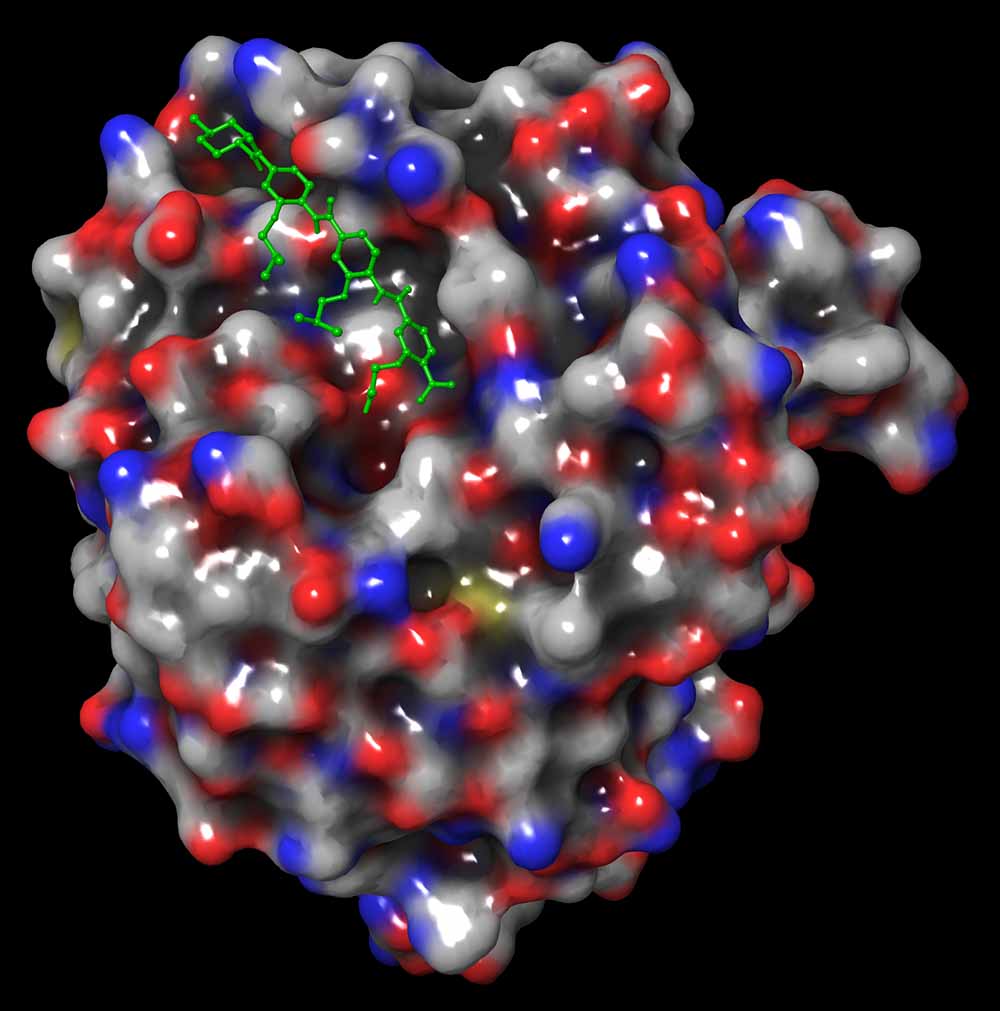
Image Source: https://news.utdallas.edu/health-medicine/new-molecule-cancer-cell-killer-2022/
The research, which was conducted in isolated cells, human cancer tissue, and human tumors produced in mice, was described in a report published online in the first week of June in the journal Nature Cancer.
Dr. Jung-Mo Ahn, a UT Dallas associate professor of chemistry and biochemistry in the School of Natural Sciences and Mathematics, has been creating tiny compounds that target protein-protein interactions in cells for over a decade. The researcher previously produced promising therapeutic candidate molecules for treatment-resistant breast cancer and prostate cancer using a structure-based rational drug design method.
In the current study, Ahn and his colleagues investigated the effects of ERX-41, a new molecule he produced, on breast cancer cells, both those with and without estrogen receptors (ERs). While there are effective treatments for people with ER-positive breast cancer, patients with triple-negative breast cancer (TNBC), which lacks estrogen, progesterone, and human epidermal growth factor 2 receptors, have limited options. TNBC is more common in women under the age of 40 and has a worse prognosis than other kinds of breast cancer.
According to Ahn, the ERX-41 compound did not harm healthy cells but destroyed tumor cells whether or not they had estrogen receptors. In fact, it was more effective at killing triple-negative breast cancer cells than it was at killing ER-positive cells.
The researchers were astonished by this at that moment and they knew it had to be targeting something in the TNBC cells other than estrogen receptors but didn’t know what it was.
Dr. Ganesh Raj, professor of urology and pharmacology at the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, and Dr. Ratna Vadlamudi, professor of obstetrics and gynecology at UT Health San Antonio, were co-corresponding authors on the ERX-41 molecule study. The chemical was synthesized with the help of Dr. Tae-Kyung Lee, a former UTD research scientist at Ahn’s Bio-Organic/Medicinal Chemistry Lab.
According to the researchers, the ERX-41 links to a biological protein termed lysosomal acid lipase A (LIPA). LIPA is present in the endoplasmic reticulum, which is a cell organelle that processes and folds proteins.
Ahn explains that to develop quickly, a tumor cell must manufacture a lot of proteins, which puts stress on the endoplasmic reticulum. Cancer cells overproduce LIPA at a far higher rate than healthy cells. By binding to LIPA, ERX-41 inhibits protein processing in the endoplasmic reticulum, causing it to bloat and eventually die.
Triple-negative breast cancer is particularly insidious — it targets women at younger ages; it’s aggressive; and it’s treatment-resistant. I’m really glad we’ve discovered something that has the potential to make a significant difference for these patients.
Dr. Jung-Mo Ahn, associate professor of chemistry and biochemistry in the School of Natural Sciences and Mathematics
The chemical was also tested in healthy mice, and no negative effects were identified.
Ahn remarked that the researchers took several years to figure out which proteins were influenced by ERX-41. That was the most difficult part. The researchers went through a lot of dead ends, but we didn’t give up.
Triple-negative Breast cancer is particularly sneaky since it strikes women at an early age, is aggressive, and is resistant to treatment. The researchers uncovered something that has the potential to help these people with hard to treat cancers significantly.
The substance was given to mice with malignant tumors similar to those found in humans, and the tumors shrank. The chemical was also found to kill cancer cells in human tissue taken from individuals who had tumors removed.
They also discovered that ERX-41 works against other malignancies with high endoplasmic reticulum stress, such as hard to treat cancers (pancreatic and ovarian tumors), as well as glioblastoma, the most aggressive and deadly primary brain tumor.
As a chemist, I am somewhat isolated from patients, so this success is an opportunity for me to feel like what I do can be useful to society.
Dr. Jung-Mo Ahn
Ahn is a co-holder of patents on ERX-41 and related substances that have been licensed to the Dallas-based startup EtiraRX, which was co-founded by Ahn, Raj, and Vadlamudi in 2018. The company has announced that clinical trials for ERX-41 will commence in the first quarter of 2023.
In addition to researchers from UT Southwestern and UT Health San Antonio, other study authors from Howard Hughes Medical Institute’s Janelia Research Campus, Northwest A&F University in China, and the Medical College of Xiamen University in China contributed to the study.
Story Source: Liu, X., Viswanadhapalli, S., Kumar, S. et al. Targeting LIPA independent of its lipase activity is a therapeutic strategy in solid tumors via induction of endoplasmic reticulum stress. Nat Cancer (2022). https://doi.org/10.1038/s43018-022-00389-8 https://news.utdallas.edu/health-medicine/new-molecule-cancer-cell-killer-2022/
Learn More About Bioinformatics:
Top Bioinformatics Books ↗
Learn more to get deeper insights into the field of bioinformatics.
Top Free Online Bioinformatics Courses ↗
Freely available courses to learn each and every aspect of bioinformatics.
Latest Bioinformatics Breakthroughs ↗
Stay updated with the latest discoveries in the field of bioinformatics.
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.






This is a topic that’s near to my heart… Cheers!