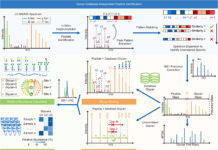
Through mass spectrometry-based proteomics, a German-Danish research team led by Matthias Mann has developed a new screening tool to identify alcohol-related liver disorders at an early stage.
Taking two or three drinks every day may put your liver in trouble. Researchers have developed a new tool that uses proteomics and machine learning to predict whether a person has alcohol-related liver disease and whether that person is in danger of disease progression. This tool is non-invasive and just as accurate as the existing state-of-the-art clinical diagnostics, if not superior. The work was co-authored by the Max Planck Institute (MPI) of Biochemistry in Germany and the Novo Nordisk Foundation (NNF) Center for Protein Research (CPR) at the University of Copenhagen and the University of Southern Denmark and was published in Nature Medicine.
Image Source: https://www.biochem.mpg.de/new-method-for-early-diagnosis-of-liver-diseases-by-proteomics
Fatty liver disease that arises due to the buildup of excess fat in the liver affects a staggering 25% of the world’s population. In fact, drinking two to three alcoholic beverages every day can harm your liver. While the fatty liver does not inherently impact your health, it does put you at risk of developing severe liver illnesses such as cirrhosis in the long run.
Even today, in order to evaluate your unique risk, a doctor must do a liver biopsy at a specialized facility. A needle is pushed through your skin and into your liver to remove a tissue sample from your liver in this type of biopsy. It’s necessary, but it’s also intrusive, and it comes with risks like bleeding. Professor Matthias Mann of the MPI of Biochemistry and the NNF-CPR has led a research team that has created a new diagnostic tool to determine whether a patient has fatty liver disease and whether they are at risk of disease development.
According to Lili Niu, who is the first author of the study and a postdoc at CPR, the detection of various types of liver damage is critical since it enhances the patients’ illness treatment. Because liver disorders often advance silently, it is critical to implement easily accessible screening programs in at-risk groups for early identification.
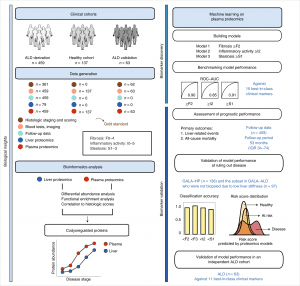
Image Source: Noninvasive proteomic biomarkers for alcohol-related liver disease
Machine learning models identified proteomics biomarkers for early detection of liver diseases
The researchers were able to detect hundreds of proteins from each patient’s blood sample in the study. A mass spectrometer, a form of very advanced weighing scale that measures molecules with extreme precision, was used to evaluate the blood samples. They employed machine learning to locate proteins connected to the existence of different types of liver injury after identifying and quantifying the proteome (total of the proteins in the sample).
The research team found three biomarker panels that can detect significant fibrosis, mild inflammatory activity, and any steatosis, all of which are multiple ways the disease can show up in the tissue, explains Lili Niu.
In a nutshell, these biomarkers are what the researchers were looking for in the blood samples since they can detect any of the aforementioned liver damage and can help forecast whether a patient is at risk of disease development.
According to Lili Niu, in the end, the researchers only needed two weeks of measurement time to analyze samples from 596 people in the main cohort and another 63 people in the validation cohort, and this throughput, paired with the proteome depth, is unparalleled.
The researchers were able to forecast the patient’s risk of alcohol-related liver damage, which affects 6% of the general population, using a simple blood sample – “with performance superior or comparable to existing state-of-the-art clinical tests,” adds Niu.
Advances in MS-based proteomics
The research team is enthusiastic about the findings of the publication since this screening procedure could make potentially life-saving testing available to the general public while causing no harm to the patients. Furthermore, this technology has the potential to be improved so that it may be used to diagnose disorders other than liver disease.
We are interested in rolling this out as a screening tool for the general population or at-risk populations, such as alcohol over-users, for early detection of liver diseases. We will continue biomarker development with mass spectrometry-based analysis for its specificity and systematic aspects, among other advantages. Further, we want to develop other assays for other diseases as well.
Matthias Mann, the corresponding author who leads the Proteomics and Signal Transduction Group at the MPI of Biochemistry and the Clinical Proteomics Group at NNF-CPR
To receive a liver biopsy or advanced imaging tests with the current diagnosis tool, you must travel to a centralized and specialist facility. With this technology, your general practitioner may only need to collect a small blood sample, explained Maja Thiele, a Clinical Professor, and Chief Physician, together with her colleagues at Odense University Hospital and the University of Southern Denmark, she was in charge of selecting and examining the 659 individuals for the study.
As per Thiele, without a doubt, this is the way things will be in the future. It’s an excellent screening tool that provides us with all of the information we require about the patients. We can evaluate liver fat, inflammation, and scar tissue in the liver with a single blood test.
Story Source: Niu, L., Thiele, M., Geyer, P.E. et al. Noninvasive proteomic biomarkers for alcohol-related liver disease. Nat Med (2022). https://doi.org/10.1038/s41591-022-01850-y
https://www.biochem.mpg.de/new-method-for-early-diagnosis-of-liver-diseases-by-proteomics
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.