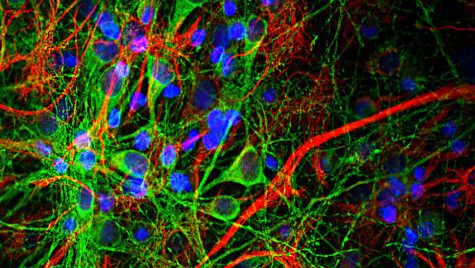
Neurons are surprisingly complicated, for something so small, not only because there are billions of them in the brain but also because their function is impacted by a variety of factors such as their structure and genetic composition.
A research group led by Daifeng Wang, professor at the University of Wisconsin–Madison, is using machine learning and artificial intelligence approaches to better understand how a variety of traits interact to influence how neurons work and behave.
The method, known as manifold learning, may aid researchers in better understanding and even predicting brain illnesses by examining unique cell features. The findings of the Wang lab were recently reported in two articles.
The researchers demonstrated that they could use manifold learning to anticipate the properties of neurons in one of the studies published in the journal Communications Biology in November 2021. They discovered that the cells could be classified based on their genes and electrophysiological behavior using current machine learning approaches, which employ computer algorithms to examine massive volumes of data and automatically generate predictions. This behavior includes neuronal electrical activity, which is critical for neuronal transmission and, ultimately, brain function.
The scientists used manifold learning to match gene expression and electrophysiological data using information from around 3,000 neurons in the mouse brain. Their purpose was to see whether there was a link between the two that could be measured.
The researchers discovered that both of these neuronal cell properties have comparable patterns — high values in one set of cells but low values in the others — and are aligned in “multi-dimensional space” or have a link with one another. This determines their so-called manifold form, which is a complicated mathematical description of the characteristics of the neurons.
“Based on this manifold shape, we found that cells can be clustered together into different groups,” says Wang.
When cells were clustered using only one feature, such as gene expression or electrophysiological, the clusters were not as well differentiated as when both characteristics were utilized together.
The researchers then wondered how genes might interact to affect cellular electrophysiology. They discovered linkages between electrophysiological properties and particular genes that affect the expression of other genes using cell clusters. Some of these genes are also implicated in immune system modulation, indicating that neuronal transmission and inflammation are linked.
Wang and his students used this information to see if they could anticipate a neuron’s electrical characteristics based on gene expression. This, according to Wang, is similar to attempting to forecast the relation between traffic patterns in a specific region of a city and the amount of take-out orders from nearby restaurants at a particular time during the day.
“Manifold learning may help researchers better understand and even predict brain disorders by looking at specific neuronal properties.”
“If you compare the traffic with the number of take-out orders from the restaurants in a particular area, they are two different things, but I believe that they share some similar patterns — like they may both have the same peak hours,” he said. “Here, we would use the manifold alignment to align the patterns between traffic (electrophysiology) and take-out order amount (gene expression) and then find the shared pattern between the two.”
You might start predicting when take-out orders would peak based on traffic data alone, or you could begin predicting the gene expression of neurons based on their electrophysiological properties, according to Wang.
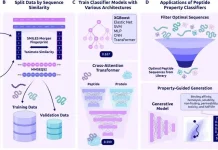
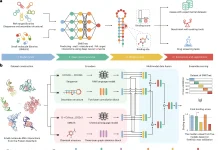
Wang’s team utilized the data they collected to inform their second research, published in January in Nature Computational Science. It provides a new and improved sort of manifold learning that overcomes the shortcomings of previous models and might aid researchers in better understanding neuron activity in health and sickness.
DeepManReg, a novel model based on gene expression and electrophysiology, enhances the prediction of neural properties. It can also include more than two types of neural characteristics and illustrate how numerous aspects interact or impact each other, making it more generalizable to various forms of cell data.
Machine learning might help these applications save time and money by reducing the amount of time and money needed to research particular aspects of the brain. Wang plans to apply the approaches to learn more about brain illnesses and diseases, despite the fact that the team’s most recent experiments were based on healthy cells.
“Basically, (we can study) how those genes are regulated to affect the electrophysiology or behaviors in diseased cells,” says Wang.
Story Source: Nguyen, N.D., Huang, J. & Wang, D. A deep manifold-regularized learning model for improving phenotype prediction from multi-modal data. Nat Comput Sci 2, 38–46 (2022). https://doi.org/10.1038/s43588-021-00185-x
Huang, J., Sheng, J. & Wang, D. Manifold learning analysis suggests strategies to align single-cell multimodal data of neuronal electrophysiology and transcriptomics. Commun Biol 4, 1308 (2021). https://doi.org/10.1038/s42003-021-02807-6; https://news.wisc.edu/researchers-are-using-machine-learning-to-understand-how-brain-cells-work/
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.