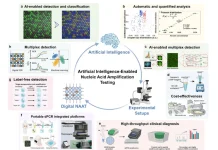
Machine learning may aid in repairing genetic damage and avoiding DNA mutations and chronic diseases.
Scientists from Massachusetts General Hospital and the National Cancer Research Centre have created novel methods for seeing DNA repair in detail, using machine learning and high-throughput microscopy, and have discovered new repair proteins. The results might open the path for novel cancer treatments.
According to the authors, “Repair of genetic damage is coordinated in the context of chromatin, so cells dynamically modulate accessibility at DNA breaks for the recruitment of DNA damage response (DDR) factors. Identifying chromatin factors with roles in DDR has mostly relied on loss-of-function screens while lacking robust high-throughput systems to study DNA repair. In this study, we have developed two high-throughput systems that allow the study of DNA repair kinetics and the recruitment of factors to double-strand breaks in a 384-well plate format.”
When a cell experiences DNA damage, such as a DNA double-strand break, the cell triggers a system known as DNA damage response, which functions as a “call to the emergency services.” Chemotherapy is designed to destroy tumor cells by generating DNA lesions, which cause cancer cells to collapse and die.
“By knowing how DNA lesions occur and how they are repaired, we will learn more about how cancer develops and how we can fight it. Any discovery in DNA repair will help develop better cancer therapies while protecting our healthy cells,” said Barbara Martínez in a press release.
The researchers employed high-throughput microscopy to see millions of photos of cells following induction of genetic damage, with the help of a machine learning analysis approach developed by the CNIO Confocal Unit. In the first step, they introduced over 300 different proteins into the cells and tested whether they interfered with DNA repair over time in a single experiment. They were able to find nine novel proteins involved in DNA repair using this method.
The new machine learning based approach provides unique opportunities for researchers to analyze and manipulate DNA repair.
The researchers analyzed hundreds of proteins quickly by applying the approach that allows direct observation of DNA repair. “An advantage is that both platforms are very versatile and can be used to discover new genes or chemical compounds that affect DNA repair,” Martínez mentioned.
Story Source: https://www.cell.com/action/showPdf?pii=S2211-1247%2821%2901676-4
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar