Researchers from the Max Planck Institute reveal the first 3D image of the CCAN protein complex and highlight basic questions about the development of artificial chromosomes.
At the heart of human cell division are hundreds of proteins. Understanding how our genetic material is reproduced and transmitted down through generations requires understanding the 3D structure of these proteins. The research team coordinated by Andrea Musacchio and Stefan Raunser from Max Planck Institute of Molecular Physiology in Dortmund has now revealed the first precise structure of CCAN, a critical protein complex for human cell division. The researchers highlight significant aspects of the complex’s 16 components using cryo-electron imaging, challenging earlier notions about how the complex recognizes the centromere, a critical area of chromosomes in cell division.
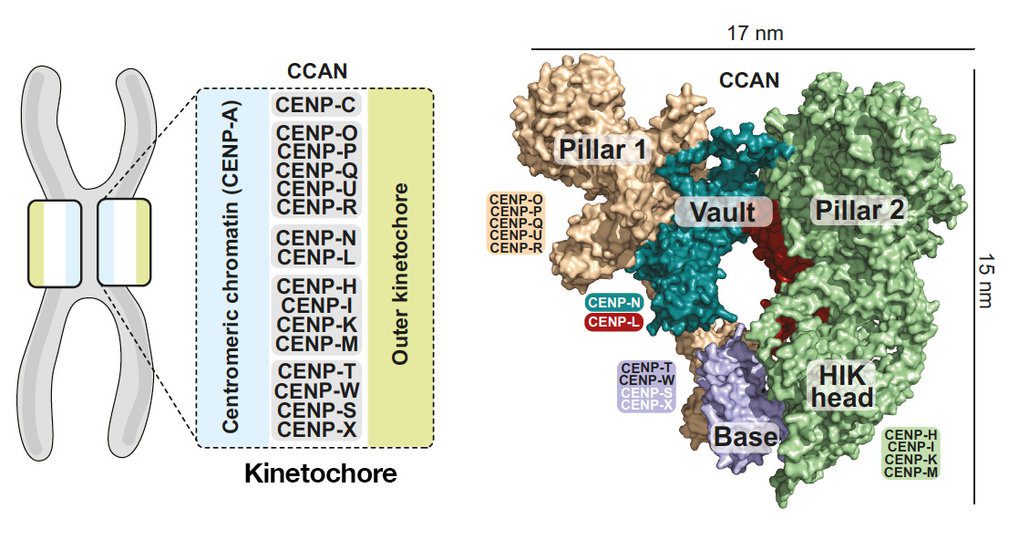
Image Source: https://www.mpi-dortmund.mpg.de/news/cell-division-key-protein
Building centromere from scratch
The centromere is a chromosomal constriction made up of DNA and proteins. The centromere is also the dock for the kinetochore, machinery made up of roughly 100 proteins that control the separation of two identical chromosomes during cell division and their delivery to daughter cells. According to a previous study, the docking of kinetochore onto the centromere occurs through the CCAN complex – the CCAN binds with the centromere protein A, the hallmark protein of the centromere. CCAN is also in charge of replacing the centromere protein A after cell division has occurred. The exact nature of CCAN’s interaction with centromere protein A is still unknown.
Understanding how CCAN recognizes and binds to the centromere could potentially lead us to build a centromere from scratch.
Andrea Musacchio
For synthetic biologists trying to create artificial chromosomes to restore missing functions or introduce new ones in cells, the centromere is a critical roadblock.
Fundamentally unanswered questions
Over 15 years ago, researchers discovered the CCAN complex. “Yet, building up a pipeline to synthesize all proteins in vitro has been a major obstacle,” told Musacchio. After achieving the first in vitro reconstruction of the human CCAN complex, Musacchio teamed up with Stefan Raunser, also from MPI Dortmund, who used cryo-electron microscopy to examine the entire CCAN protein complex.
The MPI groups were able to determine the 3D structural details of the human CCAN complex in the latest article, highlighting its unique features and consequences for a potential relationship with centromere protein A.
Contrary to what was expected, this structure does not directly recognize the centromere protein A in the standard configuration.
Andrea Musacchio
As a nucleosome, the typical unit of genetic material, the centromere protein A is most commonly packed with DNA and other proteins. The scientists now propose that the centromere protein A may be incorporated into the centromere in a new conformation, allowing it to interact with CCAN more easily. The researchers further intend to discover and prove the conditions that led to this new configuration.
Story Source: Pesenti ME, Raisch T, Conti D, Walstein K, Hoffmann I, Vogt D, Prumbaum D, Vetter IR, Raunser S, Musacchio A (2022). Structure of the human inner kinetochore CCAN complex and its significance for human centromere organization. Mol Cell. DOI: 10.1016/j.molcel.2022.04.027 https://www.mpi-dortmund.mpg.de/news/cell-division-key-protein
Dr. Tamanna Anwar is a Scientist and Co-founder of the Centre of Bioinformatics Research and Technology (CBIRT). She is a passionate bioinformatics scientist and a visionary entrepreneur. Dr. Tamanna has worked as a Young Scientist at Jawaharlal Nehru University, New Delhi. She has also worked as a Postdoctoral Fellow at the University of Saskatchewan, Canada. She has several scientific research publications in high-impact research journals. Her latest endeavor is the development of a platform that acts as a one-stop solution for all bioinformatics related information as well as developing a bioinformatics news portal to report cutting-edge bioinformatics breakthroughs.
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar
- Dr. Tamanna Anwar





