Owing to the increase in malaria infections and morbidities associated with it, an upgradation of diagnostics gold standard procedures can help elevate the disease burden. MTA-BME Morpho-dynamics Research Group from Budapest has integrated neural networks (NN), reducing the data dimension to address the limitations. High speeding the classification of RBCs (red blood cells) increases the robustness and accuracy by 98%.
Introduction
Malaria is an infectious disease with a history of the existence of more than 4 thousand years with an increased rate of infectivity and morbidity. The disease, being vector-borne and mediated by plasmodium species, still instigates a global burden inducing a threat to public health.
Infectious diseases require robust and accurate diagnostic methods to address the stages of the disease and provide the required interventions. The gold standard procedure requires assessing samples against Giemsa-stained smears followed by visualization under the light microscope. Considering the human errors during diagnosis based on handling, sample preparation can affect the diagnostic procedure. Hence, new-age diagnostics using neural networks can effectively reduce these limitations and provide one-dimensional dissections of infected RBCs.
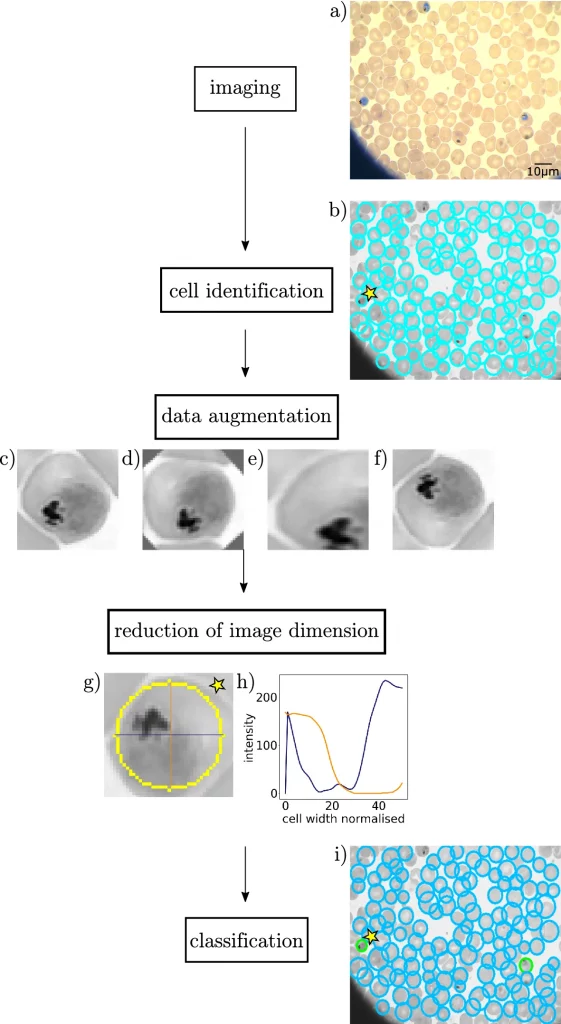
Image source: https://doi.org/10.1038/s41598-022-19601-x
Katharina preißinger et al. constructed an efficient workflow to create a neural network to analyze unpredictable imaging datasets for RBC and sort them into groups based on healthy and three stages of the disease. The trained dataset was constructed on the predictability of algorithm to reproduce results with > 95% accuracy.
The main approaches for the study included imaging data obtained from a group study utilizing data from different imaging techniques such as fluorescence microscopy 1232 images and AFM (Atomic force microscopy) 173 images, while Giemsa-stained smear data, 792 images was obtained from Abbas et al. (DOI: https://doi.org/10.1186/s13000-020-01040-9). Post imaging data, cell identification to recognize the RBCs using the Hough gradient method due to its ability to recognize erythrocytes due to circularity and large enough perimeter recognizing 95% of cells in the dataset, followed by data augmentation to training datasets for neural networks to recognize and sort RBCs into groups was done by experts manually to classify single cell RBC images into healthy, ring, trophozoite, and schizont stages, reduction of image dimension to fit parameters using asymmetric cross-sections for RBC in different cases, and finally applying a one-dimensional convolutional Neural Network (NN) to classify the cells of RBC for 4 category classification using 2D microscopic images of erythrocytes for arbitrary datasets (balanced or imbalanced).
The data augmentation of the images provided high classification accuracy across all microscopic datasets, followed by 93.34% for light microscopy, 93.2% for atomic fluorescence microscopy, and 85.9 % for fluorescence microscopy. Precision predictions also resulted in providing data for the early detection of malaria infections.
As a result, the research team employed datasets for the final performances of the neural networks based on characteristic cuts (asymmetric RBC cross-sections) optimally and reduced the dimensions of input data to fit the life stages of parasites and avoid noise and errors due to contamination. An important approach addressed by researchers was by augmenting two characteristic cuts to train and validate the neural network classifier. The results, with an average of 95%, displayed high performance and accuracy.
While there are certain limitations for neural network-based recognitions, which are the overfitting of datasets since they comprise thousands of microscopic images of healthy and infected erythrocytes, which differ in size, thus giving unbalanced data. Even post-data augmentation, researchers found the trained dataset to be too small for a 2D convolutional neural network for disease stage-specific classification.
Final Thoughts
The approach to introducing a new age of diagnostics in the era of computational biology is quite an initiative by the research group. Providing data sets into groups can help clinicians to determine the stages of infection and provide necessary interventions. The neural network model’s accuracy provides robust results that can be applied in the near future with more optimization to fit the patient’s recovery output.
Article Source: Reference Paper
Learn More:
Top Bioinformatics Books ↗
Learn more to get deeper insights into the field of bioinformatics.
Top Free Online Bioinformatics Courses ↗
Freely available courses to learn each and every aspect of bioinformatics.
Latest Bioinformatics Breakthroughs ↗
Stay updated with the latest discoveries in the field of bioinformatics.
Mahi Sharma is a consulting Content Writing Intern at the Centre of Bioinformatics Research and Technology (CBIRT). She is a postgraduate with a Master's in Immunology degree from Amity University, Noida. She has interned at CSIR-Institute of Microbial Technology, working on human and environmental microbiomes. She actively promotes research on microcosmos and science communication through her personal blog.